Abstract
Background: Chronic lymphocytic leukemia (CLL) is characterized by phenotypic and functional defects of immune cells, which often emerge into increased susceptibility to infections and autoimmunity, and also contribute to immune evasion of cancer cells. Ibrutinib is a selective inhibitor of BTK that shows activity via its direct effects on crucial survival pathways in CLL cells. In addition to its anti-neoplastic effects, ibrutinib has also shown to have immunomodulatory properties. Currently available data are mainly limited to the activity exerted by ibrutinib on conventional T cells, whereas little is known on the effects induced on other non-neoplastic immune cell populations.
Aim: The aim of this study was to evaluate the in vivo immunomodulatory effects of ibrutinib treatment on different immune compartments in patients with CLL
Methods: We included 11 CLL patients with progressive disease (PD CLL) and eligible to ibrutinib therapy. PB samples were collected from patients at baseline and after 1, 6 and 12 months of treatment with ibrutinib. For comparison, we also analyzed 5 healthy donors (HD) and 6 treatment-naïve CLL patients with stable disease (SD CLL), who were not fulfilling criteria for treatment start. We assessed the percentages and the absolute numbers of CLL cells, T cells, γδ (Vδ1 and Vγ9Vδ2) T cells, T regulatory cells (Tregs), natural killer (NK) and NK-T cells by flow cytometry using population-specific markers. The expression of activation markers and immune checkpoint receptors (i.e. CD69, PD-1, CD96, TIGIT, NKG2D) was evaluated by flow cytometry as well.
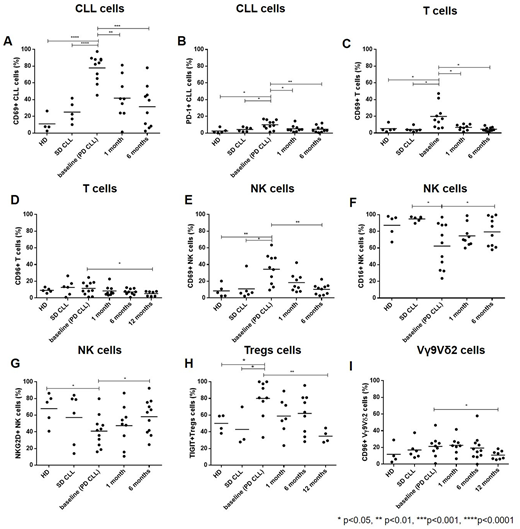
Results: Median age of patients was 69 years (range 44-75). The median lymphocyte count at study entry was 49 x 109/L (range 1,8-110) and the median number of previous treatment regimens was 2 (range 0-5). After 12 months of ibrutinib, 10 out of 11 (91%) patients achieved at least a partial response. The mean absolute number of CLL cells started to decrease by month 6 and became significantly lower than baseline value at 12 months of ibrutinib therapy. At the baseline, leukemic cells from PD CLL had significantly higher surface expression of the early activation marker CD69 and of the immune checkpoint molecule PD-1 compared to SD CLL and HD. After 6 months of treatment, the percentage of CLL cells expressing CD69 and PD-1 was normalized (reached values not significantly different from HD) (Fig 1A,B). We observed a gradual reduction of the total count of CD4+ and CD8+ T cells during ibrutinib treatment, becoming significant at 12 months. At the same timepoint, the expression of CD69, which was higher on T cells from PD CLL prior to therapy compared to SD CLL and HD, was normalized. Concurrently, a significant reduction in the surface levels of the inhibitory immune checkpoint molecule CD96 was observed (Fig 1C,D). Ibrutinib treatment had no impact on the absolute numbers of NK and NK-T cells. Compared to SD CLL and HD, NK cells from PD CLL showed a higher expression of CD69 before treatment start. In addition, they were characterized by increased levels of the immune checkpoints CD96 and TIGIT, and by reduced expression of the Fc receptor CD16, that is involved in the ADCC process, and the activating receptor NKG2D. After 6 months of treatment, the expression of CD69, CD16 and NKG2D on NK cells were restored (Fig 1E-G), whereas TIGIT and CD96 were not yet significantly modulated. Concerning the Tregs, a trend toward a reduction in the absolute number was detected after 12 months of ibrutinib treatment, compared to the baseline. The percentage of Tregs expressing the co-inhibitory molecule TIGIT was higher in PD CLL at the time of treatment start and was normalized by 12 months of ibrutinib therapy (Fig 1H). Lastly, we assessed ibrutinib effects on γδ T cells. The absolute numbers of both Vδ1 and Vγ9Vδ2 T cells remained unchanged during patients' treatment. Similar to conventional T lymphocytes, Vγ9Vδ2 T cells showed a decrease in the expression of CD96 after 12 months of ibrutinib administration (Fig 1I).
Conclusions: our data suggest that the anti-tumor activity of ibrutinib is paralleled by a valuable immunomodulatory effect, leading to a partial recovery of phenotypic alterations that are hallmarks of immune exhaustion. Further studies to investigate the ability of ibrutinib to restore the functionality of the described immune cell compartments and to explore clinical correlations are currently ongoing on an enlarged cohort of treated patients.
Mauro:abbvie: Other: board member; janssen: Other: board member. Gaidano:Gilead: Consultancy, Honoraria; Janssen: Consultancy, Honoraria; Amgen: Consultancy, Honoraria; Morphosys: Honoraria; Roche: Consultancy, Honoraria; AbbVie: Consultancy, Honoraria. Foà:ABBVIE: Other: ADVISORY BOARD, Speakers Bureau; AMGEN: Other: ADVISORY BOARD; NOVARTIS: Speakers Bureau; JANSSEN: Other: ADVISORY BOARD, Speakers Bureau; ROCHE: Other: ADVISORY BOARD, Speakers Bureau; GILEAD: Speakers Bureau; INCYTE: Other: ADVISORY BOARD; CELGENE: Other: ADVISORY BOARD, Speakers Bureau; CELTRION: Other: ADVISORY BOARD. Boccadoro:AbbVie: Honoraria; Bristol-Myers Squibb: Honoraria, Research Funding; Janssen: Honoraria, Research Funding; Amgen: Honoraria, Research Funding; Celgene: Honoraria, Research Funding; Sanofi: Honoraria, Research Funding; Mundipharma: Research Funding; Novartis: Honoraria, Research Funding. Coscia:Janssen, Karyopharm: Research Funding; Abbvie, Gilead, Shire: Honoraria, Membership on an entity's Board of Directors or advisory committees.
Author notes
Asterisk with author names denotes non-ASH members.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal